 
I recommend you to read this link, where it is easy and fun to learn about the way to calculate the number of neutrons of any element on the periodic table of elements. Hence, the Potassium number of neutrons are 20, 21 and 22. An ion of platinum has a mass number of 195 and contains 74 electrons. Since the iodine is added as a 1 anion, the number of electrons is 54 53 (1) 54. 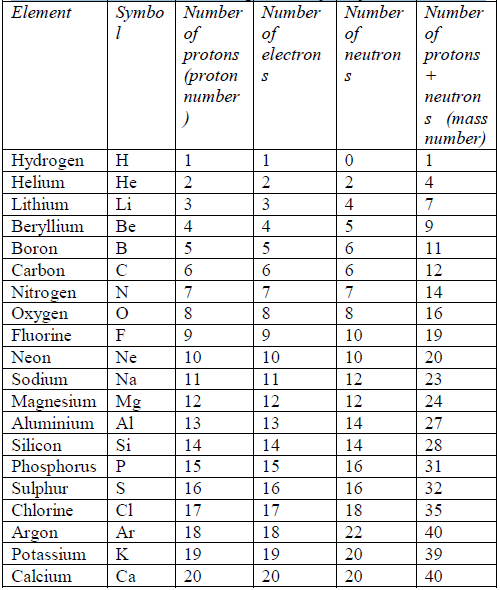
Hence, number of neutrons is 41 - 19 which is 22. Because the sum of the numbers of protons and neutrons equals the mass number, 127, the number of neutrons is 74 (127 53 74). Potassium-41 - It has 19 protons and atomic mass is 41. For example the electron configuration of potassium is Ar 4s 1. Hence, number of neutrons is 40 - 19 which is 21. 0:00 / 4:15 How to find the Number of Protons, Electrons, Neutrons for Potassium (K) Wayne Breslyn 628K subscribers 32K views 2 years ago In this video we’ll use the Periodic table and a few. The simple answer: The elements will lie in the s, p, d or f block will completely depend upon the subshell in which the last electron will enter. Potassium-40 - It has 19 protons and atomic mass is 40. Hence, number of neutrons is 39 - 19 which is 20. Potassium-39 - It has 19 protons and atomic mass is 39. An isotope is an atom of an element with the same number of protons, but differing numbers of neutrons, which means that isotopes have different atomic weights.īecause potassium has 3 isotopes, then we have to calculate for the different atomic weights of the isotopes: Number of neutrons = atomic mass number - atomic number The Bohr model shows the atom as a central nucleus containing protons and neutrons, with the electrons in circular electron shells at specific distances from the nucleus, similar to planets orbiting around the sun. Finally in order to calculate the number of neutrons of that element, you have to do some simple math: In this video we’ll use the Periodic table and a few simple rules to find the protons, electrons, and neutrons for the element Potassium (K). An early model of the atom was developed in 1913 by the Danish scientist Niels Bohr (18851962). Electrons revolve around the nucleus in a specific orbit. The only exception is hydrogen, which has only protons in its nucleus but no neutrons. Experiments by various scientists have shown that the nucleus of an atom contains protons and neutrons. 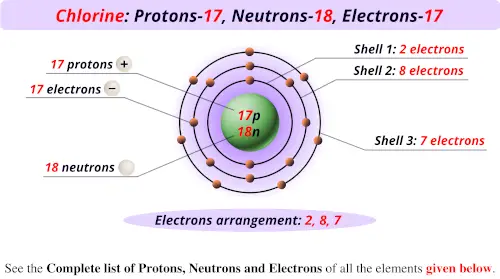
To continue with your calculation of number of neutrons, you also need to find the atomic mass of the element, which is written underneath the symbol of the element. One is the nucleus and the other is the orbit. Formula: Atomic number Number of electrons Number of protons Mass number Number of protons + Number of neutrons. Once there, you proceed to locate the “atomic number” of potassium which is the number in the upper left-hand corner of the square. Potassium has the symbol “K” and it is located in the column at the very right of the table here. In order to calculate the number of neutrons for any element in the Periodic Table of the Elements, you have to locate the element first. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
